Italy: Doctors carry out first artificial implant
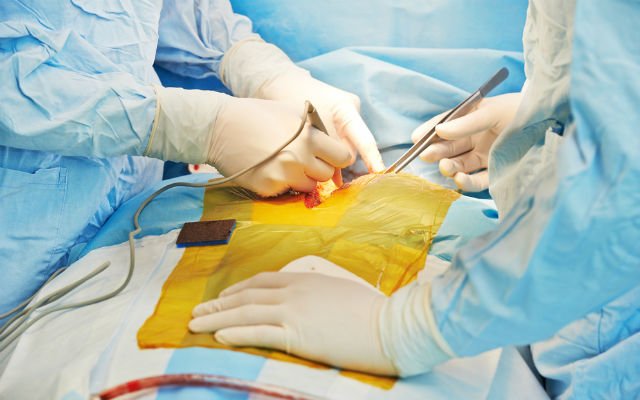
Rome: The bioprosthetic heart, developed by French company Carmat, has already been implanted into patients in clinical trial settings in the US but this operation marks the first commercial sale and use of the product.
In December 2020, Carmat received approval from the EU to sell the device as a bridge to a heart transplant for patients suffering from end-stage heart failure.
The device is made from a combination of biological and synthetic materials and was first developed in 2008.
According to Marisa De Feo, the Director of Heart Surgery at Naples’ Azienda Ospedaliera dei Colli, Monaldi hospital, the new artificial heart from Carmat compares favourably with a model by SynCardia Systems they have implanted previously.
“So far, we have implanted the SynCardia, a completely mechanical artificial heart with four mechanical valves inside, while the Carmat has four biological valves,” she said.
“It is clear already that it is much easier to manage. First of all, it is not noisy, noise being the big limitation of the other devices. The fact that it has biological valves means that there is no need to decoagulate, which reduces the risk of thrombosis and haemorrhage”.
Will we soon see bionic hearts? “Let’s say that in the next few years, we hope there will be more innovations and we hope to continue to be the first for implants,” De Feo said.





